History was made on March 24, 2015 as Retina Consultants of Hawaii director Dr. Gregg Kokame performed the first implantation of the “bionic eye,” or the ARGUS II Retinal Prosthesis System (“ARGUS II”), into the eye of a patient from Honolulu. This is the first FDA-approved device in the world that will allow patients that are completely blind to regain some vision. In addition, this was the very FIRST implantation of such a device in Hawaii and the Asia-Pacific region and only the second such surgery performed west of the Rocky Mountains. The ARGUS II was FDA-approved as a “humanitarian device” in 2013, and the first operation in the United States occurred in January 2014. Dr. Kokame’s surgery was the 20th operation performed in United States at one of only 12 centers currently approved in North America. Dr. Humayun, the inventor of the ARGUS II, met with Retina Consultants of Hawaii physicians Dr. Kokame, Dr. James Lai, and Dr. Raymond Wee at the Eye Surgery Center of Hawaii and provided guidance during the innovative four-hour operation.
ARGUS II is designed for blind patients with the most advanced form of a genetic disease known as “retinitis pigmentosa.” In a healthy retina, light enters the eye and stimulates photoreceptors (cells that respond to light). These photoreceptors then send electrical impulses to the optic nerve, which sends information to be processed by the brain. In retinitis pigmentosa, photoreceptors progressively stop responding to light, and patients can become completely blind.
ARGUS II is composed of two systems, an internal retinal implant and an external system. The retinal implant is a microelectrode array that is surgically placed on top of the retina. The external system includes a camera mounted on a pair of glasses and a battery-powered video processing unit. Visual signals enter a camera mounted on the patient’s glasses. These signals are then sent wirelessly from the camera to the microelectrode array that sits on the retina. The retinal chip then stimulates the retina, which sends electrical impulses to the optic nerve and the brain, allowing patients to see.
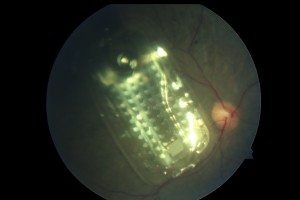
Internal system: 60-electrode array surgically placed on top of retina of blind retinitis pigmentosa patient treated in Hawaii

External System: Glasses with mounted camera (left) connected to battery-powered video-processing unit (right) wirelessly interacts with electrode in the eye

Dr. Troy Tanji (Dr. Kokame’s colleague), Dr. Kokame, and Dr. Humayan smile after a successful surgery.
The vision restored with this device will vary. Patients who are completely blind pre-operatively (in other words, cannot see any light) have been able to perceive basic shapes, lines, direction or moving objects, some depth perception, large print text, and even fireworks. Patients are not expected to be able to drive, read small text, and watch TV. Here are a few stories of what patients with the retinal chip are able to see:
- Mayo Clinic: https://newsnetwork.mayoclinic.org/discussion/bionic-eye-implant-offers-hope-of-restoring-vision/
- Fireworks: https://www.youtube.com/watch?v=8AWIJdayKow
- Duke Eye Center: https://www.youtube.com/watch?v=CiyGOUHD2nI
Also due to the groundbreaking ability to restore sight for a blind patient with retinitis pigmentosa, and to be the first surgery in the Asia Pacific, HMRI received local, national, and international attention:
Local News
- KITV 4 News: https://www.kitv.com/news/
honolulu-woman-receives- bionic-eye-implant/31998324 (includes news story video) - Hawaii News Now: https://www.hawaiinewsnow.com/
story/28617348/historic- bionic-eye-surgery-restores- womans-sight
National News
- ABC: https://abcnews.go.com/Health/
blind-hawaii-woman-bionic-eye/ story?id=29927935 (includes news story video) - Pacific Business News: https://www.bizjournals.com/
pacific/news/2015/03/19/first- retinal-implant-in-hawaii-and- asia-pacific.html
International News
- UK Daily Mail: https://www.dailymail.co.uk/
news/article-3013767/Blind- woman-72-person-Hawaii-bionic- eye-help-again.html
Drs. Kokame, Lai, and Wee are excited to have been selected as the only center in Hawaii and the Asia-Pacific region to offer this truly amazing procedure. Their hope is to be able to help patients not only from Hawaii, but also from Asia where this device is not yet available.
For more information, please call our research department at (808) 380-8060 or email research@retinahi.com.





No comments yet.